Background
Medical Device
AI Product
International Adaptation
Role
Primary UX Designer Visual Designer Ongoing (North American deployment pending FDA regulatory approval)
Timeline
Sep 27, 2025
Background
Medical Device
AI Product
International Adaptation
Role
Primary UX Designer Visual Designer Ongoing (North American deployment pending FDA regulatory approval)
Timeline
Sep 27, 2025
What It Is
What It Is
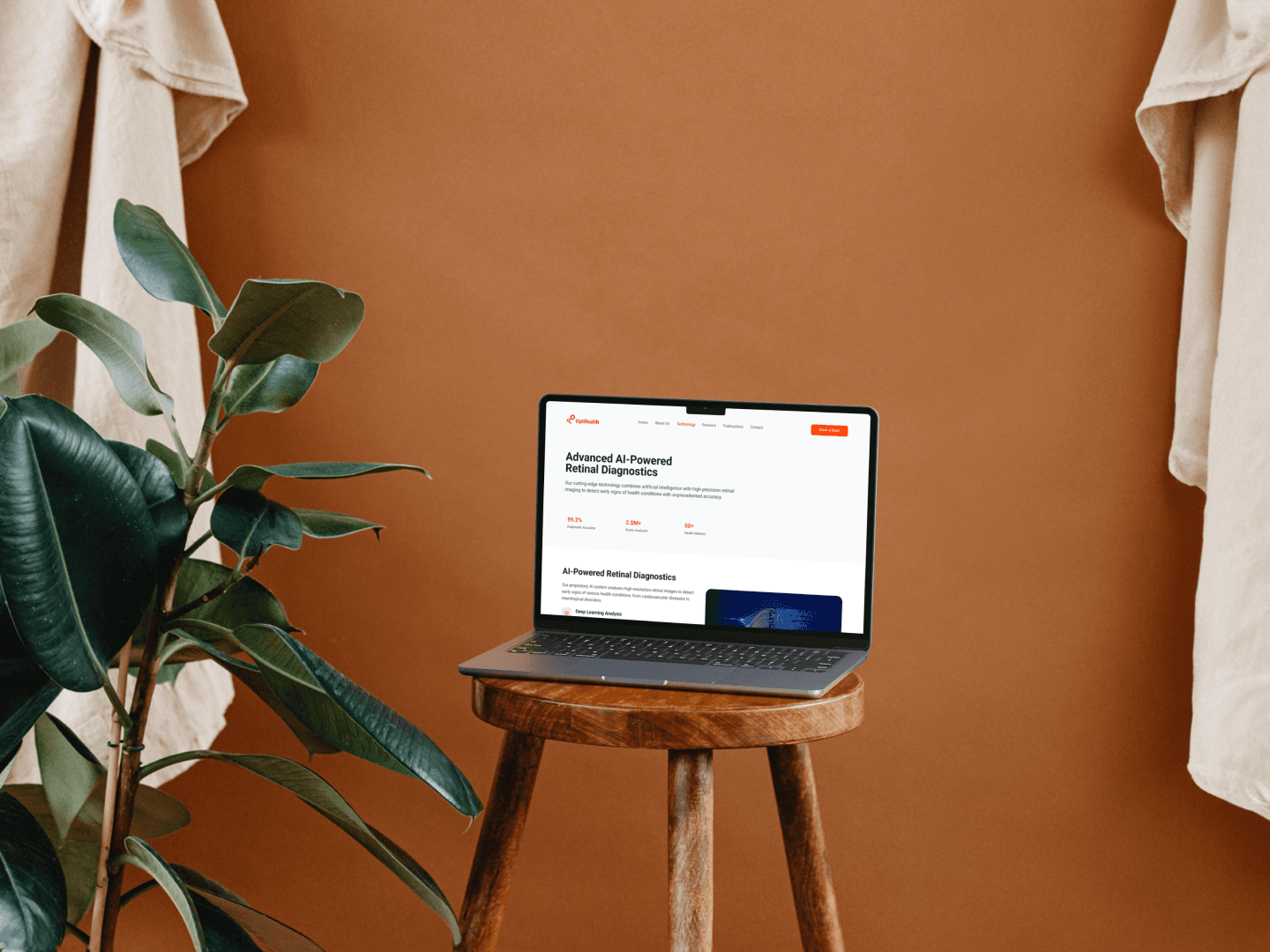
OptHealth is an AI-powered ophthalmic screening system for clinical environments. It analyzes retinal images to support physicians in early detection of diabetic retinopathy and cardiovascular risk indicators. The product is validated and live in Asian and European markets. The North American version is pending FDA-related regulatory approval. My focus was the North American adaptation: rethinking an existing product for a different regulatory environment and clinical context. Core engineering is led by the overseas team. I worked on the design and UX layer.
OptHealth is an AI-powered ophthalmic screening system for clinical environments. It analyzes retinal images to support physicians in early detection of diabetic retinopathy and cardiovascular risk indicators. The product is validated and live in Asian and European markets. The North American version is pending FDA-related regulatory approval. My focus was the North American adaptation: rethinking an existing product for a different regulatory environment and clinical context. Core engineering is led by the overseas team. I worked on the design and UX layer.
OptHealth is an AI-powered ophthalmic screening system for clinical environments. It analyzes retinal images to support physicians in early detection of diabetic retinopathy and cardiovascular risk indicators. The product is validated and live in Asian and European markets. The North American version is pending FDA-related regulatory approval. My focus was the North American adaptation: rethinking an existing product for a different regulatory environment and clinical context. Core engineering is led by the overseas team. I worked on the design and UX layer.

My Contribution
UX Design
Designed physician-facing UI: on-device interface, AI results report, and clinical workflow
Participated in cross-functional reviews with overseas product and engineering teams to align interface decisions with regulatory and workflow constraints
Regulatory Adaptation
Revised workflows and UX copy to meet FDA requirements and local clinical expectations
Shifted result language from diagnostic framing to possibility-based framing, reflecting the current approved indication scope
Reorganized the report hierarchy to serve different reader types, prioritizing clinicians who need to act quickly over those parsing raw AI parameters
Research Foundation
Competitive analysis of North American medical imaging products
FDA guidance documentation review
Cross-market product analysis comparing the Asian and European versions
Marketing and Visual Design
Produced marketing materials for Canadian market outreach
Supported preparation for the 2026 CES exhibition in Las Vegas, including localization of product-facing materials
Tools
Design: Figma, Adobe Photoshop, Illustrator, Premiere, CapCut
AI: ChatGPT, Claude, Cursor, Gemini
Collaboration: Notion, Miro, Google Drive
This project is under active development. Detailed process documentation is withheld in accordance with confidentiality agreements.
My Contribution
UX Design
Designed physician-facing UI: on-device interface, AI results report, and clinical workflow
Participated in cross-functional reviews with overseas product and engineering teams to align interface decisions with regulatory and workflow constraints
Regulatory Adaptation
Revised workflows and UX copy to meet FDA requirements and local clinical expectations
Shifted result language from diagnostic framing to possibility-based framing, reflecting the current approved indication scope
Reorganized the report hierarchy to serve different reader types, prioritizing clinicians who need to act quickly over those parsing raw AI parameters
Research Foundation
Competitive analysis of North American medical imaging products
FDA guidance documentation review
Cross-market product analysis comparing the Asian and European versions
Marketing and Visual Design
Produced marketing materials for Canadian market outreach
Supported preparation for the 2026 CES exhibition in Las Vegas, including localization of product-facing materials
Tools
Design: Figma, Adobe Photoshop, Illustrator, Premiere, CapCut
AI: ChatGPT, Claude, Cursor, Gemini
Collaboration: Notion, Miro, Google Drive
This project is under active development. Detailed process documentation is withheld in accordance with confidentiality agreements.